Research Network
The North American Registry for FMD
Beadie FMD Fact:
 FMD is a chronic vascular disease that requires lifelong care focused on medical therapy as well as periodic imaging of affected arteries
FMD is a chronic vascular disease that requires lifelong care focused on medical therapy as well as periodic imaging of affected arteries
![]()

In 2007, the Fibromuscular Dysplasia Society of America (FMDSA) decided to begin a registry to better understand the disease and its treatment. The goals of this registry are to identify patient characteristics associated with FMD, potential markers of the disease, and commonly used imaging and treatment modalities, among others.
Michigan Clinical Outcomes Research and Reporting Program (MCORRP) is the coordinating center for the FMD Registry. Beginning initially with 7 sites, the first patient was entered into the online database at the beginning of 2009.
Since funding to support the registry is very limited, these sites were chosen by a multidisciplinary committee because they treat a large enough number of patients with FMD and thus will be able to rapidly accumulate as much information as possible. As soon as additional funding becomes available, more centers will be added.
To participate in the registry you must be seen as a patient at one of the centers listed below.
Each person agreeing to participate in the registry must first sign a consent form. The consent forms are often lengthy and detailed because they are formatted similarly to those used in clinical research. It’s important to remember that the FMD registry is a data registry only – no experimental research is being conducted. Clinical data is collected in a de-identified manner and placed into a database. Clinical data includes elements such as date of diagnosis, types of tests conducted and results of these tests, past medical history, family history, subsequent clinical events and any clinical outcomes.
All of these data elements are important to determine the natural course of the disease and to determine which procedures are most effective to relieve symptoms and reduce adverse outcomes.
Since patient privacy laws were enacted in 2003, physicians must have individual patients’ consents to place data in a central registry. No names, addresses, social security numbers, or any personal information are included in the registry. Each patient is assigned a unique ID number which identifies individual patients to the treating physician. All staff involved with the registry can identify sites and patients by numbers only. Many measures are taken to secure the information including multiple firewalls, encryption codes, and de-identifiers.
Since FMD is not commonly recognized and often misdiagnosed, the only way to gather enough information to learn how to better diagnose and treat patients is to form a data registry which allows collection of many cases from multiple institutions. With large numbers of cases, physicians can create statistical models that accurately predict which tests/procedures/medications result in the best outcomes.
The initial findings from the registry were published in the medical journal Circulation in 2012. A Patient Page was also published the same year in Circulation. In 2013, an article describing the differences in the disease between men and women was published in the Journal of the American College of Cardiology. In February 2014, an AHA Scientific Statement was published in Circulation, highlighting best practices in the care of FMD and avenues for future research. Researchers have also been able to present many abstracts and posters at national meetings with information gained from the registry. To date, almost 40 manuscripts, abstracts and posters have been published and presented with data from the patient registry.
Thank you to the FMD patients who have participated and to those who donate, together we can make a difference. As we continue to fund the registry we will continue to learn and find answers.
As of January 2026, 22 sites are active, and over 5125 patients have participated in the registry
Some registry centers have additional physicians not listed below; when scheduling, you may request the first available appointment.
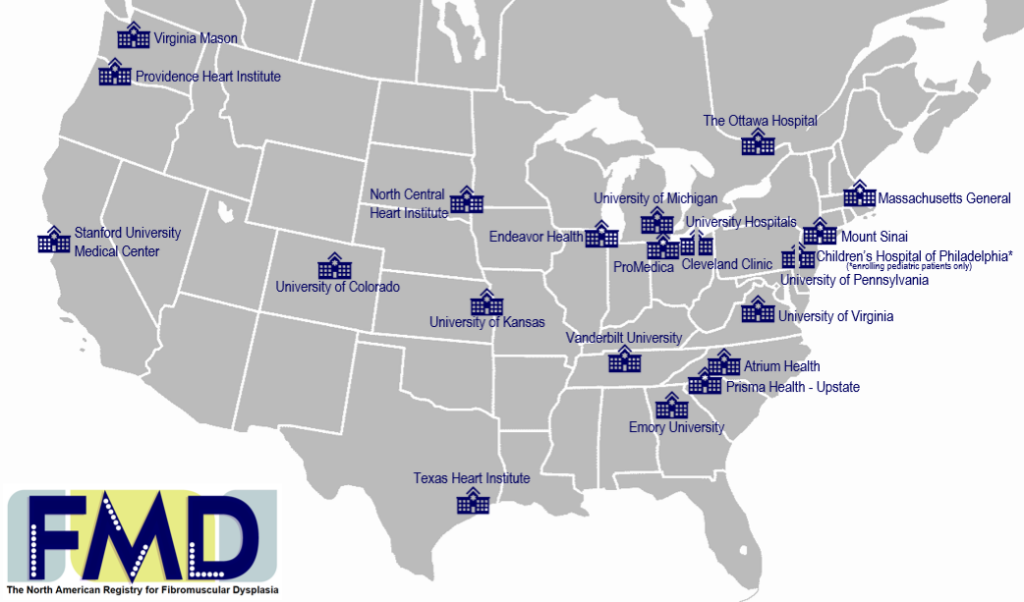
FMD Contacts:
Eri Fukaya, MD, PhD
Stanford Vascular Clinics,
Stanford, CA
(650) 725-5227
Kevin Rogers, MD
University of Colorado
Aurora, CO
(720) 848-5300
Bryan J. Wells, MD
Emory University
Atlanta, GA
(404) 686-7878
(404) 778-8440
Kambiz Zorriasateyn, MD FACP RPVI
Northshore Cardiovascular Institute
1000 Central Street, Suite 730 Evanston, IL, 60201
847-663-8050
Kamal Gupta, MD
University of Kansas
Kansas City, KS
(913) 588-9700
Gaurav Parmar, MD
Massachusetts General Hospital,
Boston, MA
(617) 726-2256
James B. Froehlich, MD, MPH
University of Michigan
Ann Arbor, MI
(888) 287-1082
Jeffrey W. Olin, DO, FACP, FACC
Daniella Kadian-Dodov, MD
Mount Sinai School of Medicine
New York, NY
(212) 241-9454
(212) 427-1540
Esther Kim MD
Dr. Aditya Sharma
Sanger Heart & Vascular Institute
Atrium Health
Charlotte, NC
(704) 373-0212
Natalia Fendrikova Mahlay, MD
Cleveland Clinic Heart and
Vascular Institute, Cleveland, OH
(216) 444-4420
Heather L. Gornik, MD
University Hospitals Cleveland
Medical Center, Cleveland, OH
(216) 844-3800
John Fish, MD
Associate Professor,
Department of Medicine,
University of Toledo, Ohio
419-291-2003
Dr. Alejandro Perez
Providence Heart Institute
Portland, OR
503-962-1000
Debbie L. Cohen, MD
University of Pennsylvania
Philadelphia, PA
(215) 662-2638
Kevin Meyers, MD
Children’s Hospital of Philadelphia
Philadelphia, PA
(215) 590-2449
Andrew Bryant Dicks, MD
Prisma Health – Upstate
Greenville, SC
(864) 454-8272
J. Michael Bacharach, MD
North Central Heart
Sioux Falls, SD
(605) 977-5000
Aaron Aday, MD, MSc,
Vanderbilt University Medical Center
Nashville, TN
(615) 322-2318
Dr. Stephanie Coulter
The Texas Heart Institute
Houston, TX
(713) 600-9213
Dr. Randy Ramcharitar
University of Virginia
Health System,
Charlottesville, VA
(434) 243-1000
Dr. Mehrdad Farid
Virginia Mason,
Seattle, WA
206-223-6637
![]() Dr. Swapnil Hiremath
Dr. Swapnil Hiremath
Renal Hypertension Clinic
Division of Nephrology
The Ottawa Hospital
Ottawa, ON, Canada
Phone 613-738-8400
ext 82480
*Referral required
Beadie FMD Fun Fact:
 FMD is a chronic vascular disease that requires lifelong care focused on medical therapy as well as periodic imaging of affected arteries
FMD is a chronic vascular disease that requires lifelong care focused on medical therapy as well as periodic imaging of affected arteries
![]()
Manuscripts
- Imad Bagh, MD; Jeffrey W. Olin, DO; James B. Froehlich, MD; Eva Kline-Rogers, MS, NP; Bruce Gray, DO; Esther S. H. Kim, MD; Aditya Sharma, MD; Ido Weinberg, MD; Bryan J. Wells, MD; Xiaokui Gu, MA; Heather L. Gornik, MD. Association of Multifocal Fibromuscular Dysplasia in Elderly Patients With a More Benign Clinical Phenotype. JAMA Cardiology. June 20, 2018. doi:10.1001/jamacardio.2018.1638.
- Joshua A. Beckman, MD; Mark A. Creager, MD. People Have the Power. Fibromuscular Dysplasia Complications. Journal of the American College of Cardiology. 2016;68(2);186-188.
- Daniella Kadian-Dodov, MD, Heather L. Gornik, MD, MHS, Xiaokui Gu, MA, James Froehlich, MD, MPH, J. Michael Bacharach, MD, MPH, Yung-Wei Chi, DO, Bruce H. Gray, DO, Michael R. Jaff, DO, Esther S.H. Kim, MD, MPH, Pamela Mace, RN, Aditya Sharma, MBBS, Eva Kline-Rogers, MS, RN, NP, Christopher White, MD, Jeffrey W. Olin, DO. Dissection and Aneurysm in Patients With Fibromuscular Dysplasia : Findings From the U.S. Registry for FMD. Journal of the American College of Cardiology. 2016;68(2);176-185
- O’Connor S, Gornik HL, Froehlich JB, Gu X, Gray BH, Mace PD, Sharma A, Olin JW, Kim ES. Smoking and Adverse Outcomes in Fibromuscular Dysplasia: US Registry Report. J Am Coll Cardiol. 2016;67(14):1750-1.
- Kuck CE, Heidt ST, Kline-Rogers E. “Rare” Diseases: Motivated Patients Make the Difference. Int J Cardiol. 2016;208:95-96.
- Green R, Gu X, Kline-Rogers E, Froehlich J, Mace P, Gray B, Katzen B, Olin J, Gornik HL, Cahill AM, Meyers KE. Differences between the pediatric and adult presentation of fibromuscular dysplasia: results from the US Registry. Pediatric Nephrology 2016;31(4): 641-50.
- Weinberg I, Gu X, Giri J, Kim SE, Bacharach MJ, Gray BH, Katzen BT, Matsumoto AH, Chi YW, Rogers KR, Froehlich J, Olin JW, Gornik HL, Jaff MR. Anti-platelet and anti-hypertension medication use in patients with fibromuscular dysplasia: Results from the United States Registry for Fibromuscular Dysplasia. Vasc Med. 2015;20(5):447-453.
- O’Connor SC, Gornik HL. Recent Developments in the Understanding and Management of Fibromuscular Dysplasia. J Am Heart Assoc. 2014;3:e001259.
- Sharma AM, Kline B. The United States Registry for Fibromuscular Dysplasia: New Findings and Breaking Myths. Tech Vasc Interventional Rad. 2014;17(4):258-263.
- O’Connor SC, Olin JW, Gornik HL. Top 8 Lessons Learned From the US Registry for FMD: Understanding the impact of registry data on clinical practice. Endovascular Today. 2014;2:32-37.
- Kim ESH, Olin JW, Froehlich JB, Gu X, Bacharach JM, Gray BH, Jaff MR, Katzen BT, Kline-Rogers E, Mace P, Matsumoto AH, McBane RD, White CJ, Gornik HL. Clinical Manifestations of Fibromuscular Dysplasia Vary by Patient Sex: A Report of the United States Registry for Fibromuscular Dysplasia. JACC. 2013;62(21):2026-2028.
- Olin JW, Froehlich J, Gu X, Bacharach M, Eagle K, Gray B, Jaff MR, Kim E, Mace P, Matsumoto AH, McBane R, Kline-Rogers E, White C, Gornik HL. The United States Registry for Fibromuscular Dysplasia: results in the first 447 patients. Circulation. 2012;125(25):3182-3190.
Abstracts
- Swan K, Gu X, Kline-Rogers E, Wells BJ, Repack A, Krallman R, Kim ES, Olin JW, Gornik HL, Mace PD, Froehlich JB, Bumpus SM. Prevalence of Headaches in Patients with Fibromuscular Dysplasia. 2017;10:A099.
- Bumpus SM, Heidt ST, Krallman R, Kline-Rogers E. Single Site Lifestyle Survey in FMD Patients Suggests Improvement Over Time. Circ Cardiovasc Qual Outcomes. 2017;10:A231.
- Kim JB, Heidt ST, Gu X, Gornik HL, Olin JW, Mace PD, Kline-Rogers E, Gray BH, Sharma A, Froehlich JB. Differences in Presenting Symptoms of Fibromuscular Dysplasia Patients with Family History of Fibromuscular Dysplasia, Arterial Aneurysm, Dissection, or Sudden Death. Circulation. 2016;9:A145.
- Rebecca Green BA, Xiaokui Gu MA, Eva Kline-Rogers MS, RN, James Froehlich MD, Pamela Mace RN, Jeffrey W Olin DO, Heather Gornik MD, Kevin E Meyers MD. The US Registry for Fibromuscular Dysplasia (USFMD): Results in the First 24 Patients < 18 Years. PAS 2015.
- Moore EK, Gu X, Olin JW, Froehlich JB, Bacharach JM, Jaff MR, Katzen BT, Kline-Rogers E, Mace PD, Matsumoto AH, McBane RD, White CJ, Kim ESH, Gornik H, Gray BH. Registry findings of fibromuscular dysplasia of the mesenteric arteries. SVM 2014.
- O'Connor S, Olin JW, Gu X, Bacharach JM, Chi Y, Froehlich JB, Gray B, Kim E, Kline-Rogers E, Mace P, McBane R, Sharma A, Gornik H. Fibromuscular dysplasia is a morbid disease with low associated mortality: A report of the United States Registry for Fibromuscular Dysplasia. SVM 2014.
- Mahmood RZ, Olin J, Gu X, Kline-Rogers E, Froehlich JB, Bacharach JM, Chi Y, Gray B, Jaff M, Katzen B, Kim E, Mace P, McBane R, Sharma A, White C, Gornik H. Unraveling Pulsatile Tinnitus in FMD: A Report of the United States Registry for Fibromuscular Dysplasia. ACC 2014.
- Kadian-Dodov D, Gornik H, Gu X, Froehlich JB, Bacharach JM, Gray B, Jaff M, Katzen B, Kim E, Laird J, Mace P, Matsumoto A, McBane R, Kline-Rogers E, White C, Olin J. Aneurysm and Dissection in Fibromuscular Dysplasia: Findings from the United States Registry for FMD. ACC 2014
- O'Connor S, Gornik H, Olin JW, Froehlich JB, Gu X, Bacharach JM, Gray BH, Katzen B, Sharma A, McBane RD, Jaff M, Chi Y, Mace PD, Kim ESH. Association between Smoking and the Natural History of Renal and Cerebrovascular Fibromuscular Dysplasia: a Report of the United States Registry for FMD. AHA 2014.
- Gavin J, Gu X, Olin JW, Gornik HL, Kim E, Gray BH, Jaff MR, McBane RD, Katzen BT, White CJ, Mace PD, Kline-Rogers E, Froehlich JB. Prevalence of Arterial Aneurysms in Patients with Fibromuscular Dysplasia. ACC 2013.
- Kim E, Olin JW, Froehlich JB, Gu X, Bacharach M, Gray BH, Jaff MR, Katzen BT, Kline-Rogers E, Mace PD, Matsumoto AH, McBane RD, White CJ, Gornik HL. Clinical Manifestations of Fibromuscular Dysplasia Vary by Patient Sex. ACC 2013.
- Weinberg I, Gu X, Giri J, Rogers K, Jaff MR. Patterns of Medication Use in 615 Patients with Fibromuscular Dysplasia. SVM 2013.
- Gavin J, Gu X, Gornik HL, Olin JW, Kim E, Mace PD, Gray BH, Bachrach M, Jaff MR, McBane RD, Matsumoto AH, Kline-Rogers E, Froehlich JB. Factors Associated with Delay in Diagnosis of Patients with Fibromuscular Dysplasia. SVM 2013.
- Verma R, Gu X, Kline-Rogers E, Gornik H, Olin J, Kim E, Bruenger P, Mace P, Froehlich JB. Understanding Patient Characteristics and Clinical Significance of Headaches in Patients with Fibromuscular Dysplasia. AHA 2013.
- Poloskey SL, Olin JW, Froehlich JB, Gu X, Bacharach M, Gray B, Grise MA, Jaff MR, Kim E, Kline-Rogers E, Mace P, Matsumoto A, McBane R, Rooke TW, Gornik HL. Physical Examination Findings in Fibromuscular Dysplasia. ACC 2012.
- Olin JW, Gu X, Froehlich JB, Bacharach M, Eagle K, Gray B, Grise MA, Jaff MR, Kim E, Kline-Rogers E, Mace PD, Matsumoto A, McBane R, Gornik HL. Peripheral Artery Dissection in Patients with Fibromuscular Dysplasia. ACC 2012.
- Ralko A, Gu X, Kline-Rogers E, Gornik HL, Olin JW, McBane R, Bacharach M, Kim E, Mace P, Grise M, Gray B, Froehlich J. Diagnostic and Follow-Up Imaging for Fibromuscular Dysplasia. ACC 2012.
- Olin JW, Froehlich JB, Gu X, Bacharach JM, Gray B, Grise MA, Kim SH, Kline-Rogers E, Mace PD, McBane R, Gornik HL.Clinical Features and Presenting Symptoms of Fibromuscular Dysplasia: A Report of the Fibromuscular Dysplasia Patient Registry. ACC 2011.
- Gornik HL, Froehlich JB, Gu X, Bacharach JM, Gray B, Grise MA, Kim SH, Kline-Rogers E, Mace PD, McBane R, Olin JW. Morbidity, Vascular Events and Interventional Therapy for Fibromuscular Dysplasia: A: A Report of the Fibromuscular Dysplasia Patient Registry. ACC 2011.
Posters
Clinical Manifestations of Fibromuscular Dysplasia Vary by Patient Sex, presented at ACC 2013
Download PDFFactors Associated with Delay in Diagnosis of Patients with Fibromuscular Dysplasia, presented at SVM 2013
Download PDFPhysical Examination Findings in Fibromuscular Dysplasia, presented at
ACC 2012
Prevalence of Arterial Aneurysms in Patients with Fibromuscular Dysplasia, presented at ACC 2013
Download PDFDiagnostic and Follow-Up Imaging for Fibromuscular Dysplasia, presented at
ACC 2012
Morbidity, Vascular Events and Interventional Therapy for Fibromuscular Dysplasia: A Report of the Fibromuscular Dysplasia Patient Registry, presented at ACC 2011
Download PDF
Beadie Fun Fact
Whereas FMD is highly prevalent among patients with SCAD, coronary dissection is an uncommon occurrence among patients with FMD
Other FMD Research Findings
First International Consensus on the diagnosis and management of fibromuscular dysplasia
First International Consensus on the diagnosis and management of fibromuscular dysplasia Heather L Gornik (Co-Chair), Alexandre Persu (Co-Chair), David Adlam, Lucas S Aparicio, Michel Azizi, Marion Boulanger, Rosa Maria Bruno, […]
Read MoreGenomics of Fibromuscular Dysplasia
Genomics of Fibromuscular Dysplasia This study was conducted by Alexandre Persu and his team of FMD researchers out of UCL located in Brussels. Abstract: Fibromuscular Dysplasia (FMD) is “an idiopathic, […]
Read MoreFibromuscular Dysplasia (FMD)
Fibromuscular Dysplasia (FMD) Marianne H Khoury, Heather L Gornik Vascular Medicine. Vol 22, Issue 3, 248 – 252. April 3, 2017 DOI: https://doi.org/10.1177/1358863X17700716 Fibromuscular dysplasia (FMD) is an uncommon vascular disease […]
Read MoreLoss-of-Function Mutations in YY1Ap1 Lead to Grange Syndrome and a Fibrouscular Dysplasia-Like Vascular Disease
Loss-of-Function Mutations in YY1Ap1 Lead to Grange Syndrome and a Fibrouscular Dysplasia-Like Vascular Disease Dong-chuan Guo13, Xue-Yan Duan13, Ellen S. Regalado, Lauren Mellor-Crummey, Callie S. Kwartler, Dong Kim, Kenneth Lieberman, […]
Read MoreStigma, Fear, and Acceptance: Three Phases of the Fibromuscular Dysplasia Experience.
Stigma, Fear, and Acceptance: Three Phases of the Fibromuscular Dysplasia Experience. Bumpus SM, Krallman R, Heidt S, Kline-Rogers E. JSM Atherosclerosis. 2017;2(1):1021. Fibromuscular Dysplasia (FMD) is an arteriopathy that can […]
Read More
Beadie Fun Fact
The most common type of Fibromuscular Dysplasia, multifocal FMD, is an artery that has the appearance of a string of beads on imaging
Other FMD Related Research Studies
University of Michigan
University of Michigan Santhi K. Ganesh, MD and colleagues at the University of Michigan are conducting research on the genetic basis of arterial dysplasia. If you agree to participate we […]
Read MoreMount Sinai Heart Center for Fibromuscular Dysplasia Care and Research
Mount Sinai Heart Center for Fibromuscular Dysplasia Care and Research The prevalence, cause, prognosis, and optimal treatment of fibromuscular dysplasia (FMD) is not known. Despite an improved clinical understanding of […]
Read MoreNACAD Registry – Primary Investigator, Dr Jackie Saw
NACAD Registry – Primary Investigator, Dr Jackie Saw The Non-Atherosclerotic Coronary Artery Disease (NACAD) Registry is an ongoing patient registry approved by the UBC research ethics board for patients with […]
Read MoreMayo Clinic
Mayo Clinic The Mayo Clinic Spontaneous Coronary Artery Dissection (SCAD) Research Program is part of an innovative multidisciplinary collaborative research and clinical practice initiative formed in 2010. The goal of […]
Read MoreThe European/International FMD Registry and Initiative (FEIRI)
The European/International FMD Registry and Initiative (FEIRI) The European/International FMD Registry and Initiative (FEIRI) Is coordinated by Prof Alexandre Persu (Clinique Universitaires, Université Catholique de Louvain, Brussels, Belgium) with the […]
Read More

 Since ancient times and across many cultures, the tree has long been a symbol of many things, including wisdom, protection, strength, inter-connectivity, and life itself. We’ve chosen the tree to represent the Fibromuscular Dysplasia Society of America for these reasons, and because the tree’s branches and roots signify the arteries affected by this disease. The tree’s branches, reaching upward and outward, provide protection and shelter, and represent our mission to help those who’ve been diagnosed with FMD. Trees, strengthened by their continuously spreading roots, symbolize how as we grow together we are stronger and more resilient. As with its growth from a delicate sapling to a giant robust tree, and as its branches and roots grow and spread, the tree symbol represents how we’ve evolved and continue to promote research, education and patient support throughout the years. Together we can achieve much, as we strive to reach for the sky.
Since ancient times and across many cultures, the tree has long been a symbol of many things, including wisdom, protection, strength, inter-connectivity, and life itself. We’ve chosen the tree to represent the Fibromuscular Dysplasia Society of America for these reasons, and because the tree’s branches and roots signify the arteries affected by this disease. The tree’s branches, reaching upward and outward, provide protection and shelter, and represent our mission to help those who’ve been diagnosed with FMD. Trees, strengthened by their continuously spreading roots, symbolize how as we grow together we are stronger and more resilient. As with its growth from a delicate sapling to a giant robust tree, and as its branches and roots grow and spread, the tree symbol represents how we’ve evolved and continue to promote research, education and patient support throughout the years. Together we can achieve much, as we strive to reach for the sky.